nitride
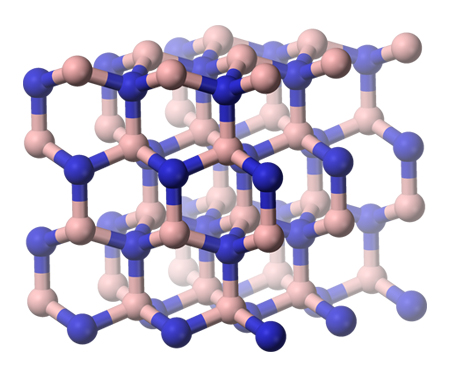
In chemistry, a nitride is a compound of nitrogen where nitrogen has a formal oxidation state of −3. Nitrides are a large class of compounds with a wide range of properties and applications.
The nitride ion, N3−, is never encountered in protic solution because it is so basic that it would be protonated immediately. Its ionic radius is estimated to be 140 pm.
Like carbides, nitrides are often refractory materials owing to their high lattice energy which reflects the strong attraction of “N3−” for the metal cation. Thus, titanium nitride and silicon nitride are used as cutting materials and hard coatings. Hexagonal boron nitride, which adopts a layered structure, is a useful high-temperature lubricant akin to molybdenum disulfide. Nitride compounds often have large band gaps, thus nitrides are usually insulators or wide bandgap semiconductors; examples include boron nitride and silicon nitride. The wide band gap material gallium nitride is prized for emitting blue light in LEDs. Like some oxides, nitrides can absorb hydrogen and have been discussed in the context of hydrogen storage, e.g. lithium nitride.